CIPARS
General
The
Public Health Agency of Canada coordinates the Canadian Integrated Program for
Antimicrobial Resistance Surveillance (CIPARS). CIPARS monitors AMU in humans, animals,
and crops, as well as AMR in select bacteria from humans, animals, and food.
CIPARS has active surveillance of AMU on volunteer sentinel farms for
grower-finisher pigs, broiler chickens, and turkeys. Farm-level surveillance
started in 2006 for grower-finisher pigs and expanded in 2013 to broiler
chickens and turkeys (with subsequent additional regional expansion). Data and
sample collection is underway on feedlot beef and dairy farms marking the
beginning of ongoing surveillance in these sectors.
Data collection
For
CIPARS, AMU data are collected from a sample of farms using a questionnaire.
The number of farms sampled each year is approximately 90-100 for pigs, 140 for
chickens, and 100 for turkeys. The denominator represents the number of animals
at risk for a single grow out period of the production stage covered by the
questionnaire. In addition to quantitative AMU data, CIPARS collects extensive
contextual information about the farms, including information about
vaccinations, biosecurity, and other routine farm management practices.
Selection criteria:
Given the limitations on the sample size of farms, inclusion/exclusion criteria were applied to ensure representativeness and internal validity of the data.
Swine: For inclusion, herds must be Canadian Quality Assurance (CQA® – a HACCP-based on-farm food safety certification program) validated, produce more than 2000 market pigs per year, and be representative of the characteristics and geographic distribution of herds in the veterinarian’s swine practice. Exclusion criteria include 1) being regarded as organic, 2) animals having been fed edible residual material or 3) the animals were raised on pasture.
Broiler chickens: The inclusion criteria involve being ‘Safe, Safer, Safest™’ compliant and a quota-holding broiler operation. Selected flocks are reflective of the veterinarian’s practice profile, representative of hatcheries supplying chicks, and representative of feed mills supplying feeds in the province/region. Exclusion criteria include being a pasture, backyard or small-sized farm
Turkeys: Inclusion and exclusion criteria are similar to those for broiler chickens with the modification that enrolled farms comply with Turkey Farmers of Canada’s On-farm Food Safety Program©.
Animal subcategories: AMU information for pigs is collected from the grower-finisher production stage. For poultry, the data are collected from the broiler/grow out stage, but the questionnaire also requests information (if known) about AMU at the hatchery-level. For turkeys, data are collected from the different weight categories (broilers, light and heavy hens and light and heavy toms) for the grow-out period.
Input: Data are manually provided to CIPARS by the veterinarians who administer the questionnaire to the producers. The data are entered into a customized database. The database incorporates automated data validity checks to identify erroneous data or data entry errors; in the case of erroneous data, the veterinarians are contacted for clarification.
Selection criteria:
Given the limitations on the sample size of farms, inclusion/exclusion criteria were applied to ensure representativeness and internal validity of the data.
Swine: For inclusion, herds must be Canadian Quality Assurance (CQA® – a HACCP-based on-farm food safety certification program) validated, produce more than 2000 market pigs per year, and be representative of the characteristics and geographic distribution of herds in the veterinarian’s swine practice. Exclusion criteria include 1) being regarded as organic, 2) animals having been fed edible residual material or 3) the animals were raised on pasture.
Broiler chickens: The inclusion criteria involve being ‘Safe, Safer, Safest™’ compliant and a quota-holding broiler operation. Selected flocks are reflective of the veterinarian’s practice profile, representative of hatcheries supplying chicks, and representative of feed mills supplying feeds in the province/region. Exclusion criteria include being a pasture, backyard or small-sized farm
Turkeys: Inclusion and exclusion criteria are similar to those for broiler chickens with the modification that enrolled farms comply with Turkey Farmers of Canada’s On-farm Food Safety Program©.
Animal subcategories: AMU information for pigs is collected from the grower-finisher production stage. For poultry, the data are collected from the broiler/grow out stage, but the questionnaire also requests information (if known) about AMU at the hatchery-level. For turkeys, data are collected from the different weight categories (broilers, light and heavy hens and light and heavy toms) for the grow-out period.
Input: Data are manually provided to CIPARS by the veterinarians who administer the questionnaire to the producers. The data are entered into a customized database. The database incorporates automated data validity checks to identify erroneous data or data entry errors; in the case of erroneous data, the veterinarians are contacted for clarification.
Analysis
Analysis
is conducted using count-based, weight-based and dose-based units of
measurement and indicators. Both Canadian and EMA[1]
standards for the average daily dose are used, though primary reporting is using
the Canadian standards. The animal weights to determine the kg animal at risk
of treatment are from EMA[2]
or specific to Canada, based on input from the Canadian industry.
Benchmarking & Reporting
Benchmarking: CIPARS currently does not conduct farm-level benchmarking.
Reporting: Annual results are communicated to the farm industries and veterinarians. CIPARS hosts a multi-commodity stakeholder webinar during the Global Antibiotic Awareness Week each year. When emerging issues are identified, CIPARS communicates these findings via surveillance bulletins and/or ad hoc meetings with relevant industry sectors, veterinary groups and government agencies. CIPARS also presents findings at local, national, and international fora and publishes select findings in peer-reviewed journals..Stewardship
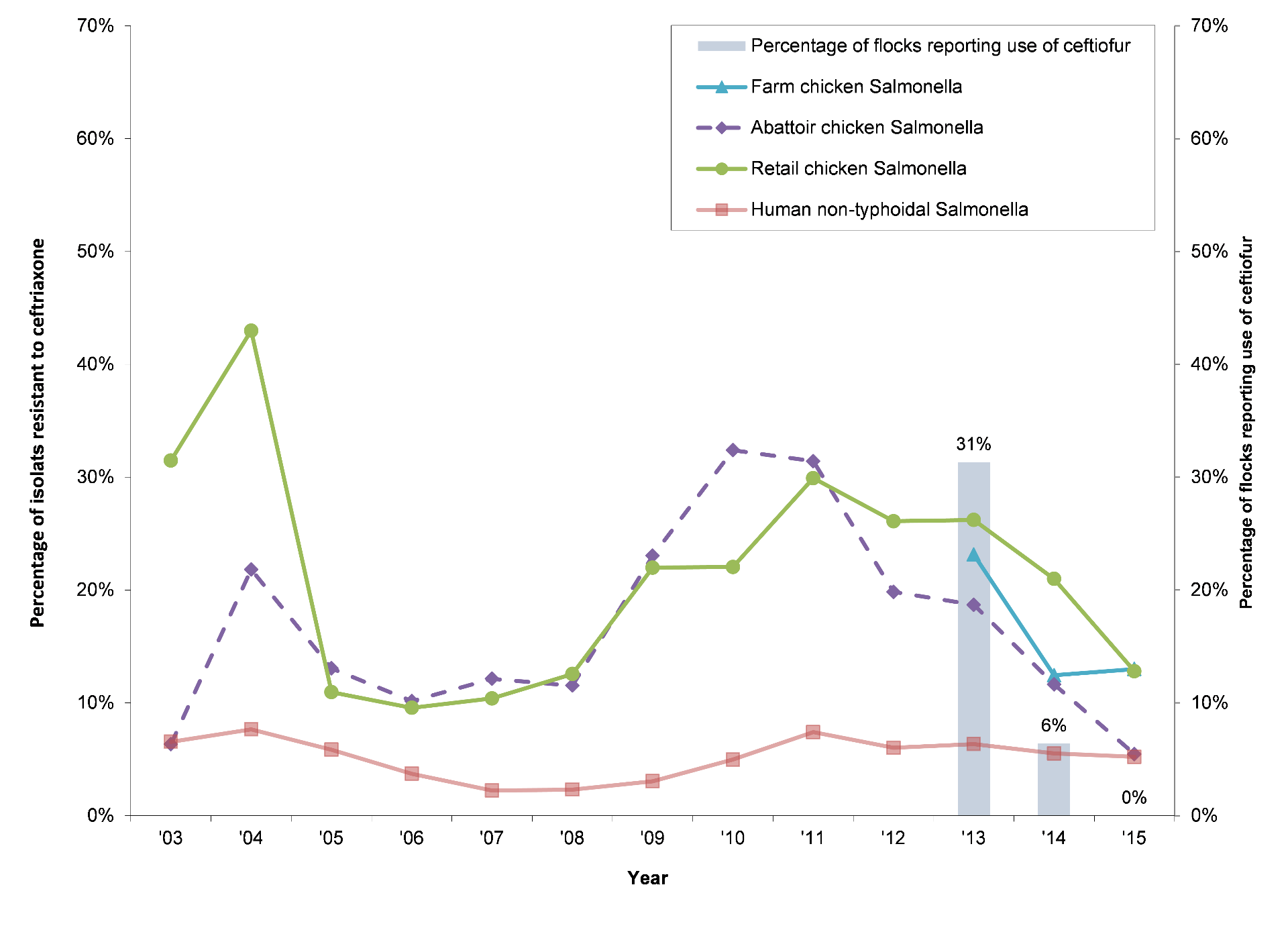
CIPARS
farm-level surveillance indicated that a change in antimicrobial use policy on
broiler chicken farms across Canada appears to be having the desired goal of
reducing use of critically important antimicrobials, in particular the use of 3rd
generation cephalosporins (Figure 1). Similarly, industry lead initiatives in
the swine production have resulted in decreasing trends in the frequency and
quantity of antimicrobials used since 2014.