Sanitel-Med
General
Data collection
Animal subcategories: Pigs: sows, finishers, gilts, weaners and sucklers; veal calves; broilers and laying hens; in cattle: calves 0-8 months, young stock 8-24 months, adult cattle.
Input: The veterinarians are legally obliged to report AMU: they have to register what they prescribe, deliver at the farms or dispense to the animals. There are four data-lock points for veterinarians: 15 April, 15 July, 15 October, 15 January. The farmers can check and validate the registrations but can also wait for automatic validation at the farmer’s data-lock points (30 April, 31 July, 31 October, 31 January). The farmers can change the quantity antimicrobials registered or can refuse the registrations. Changing data after the data lock points is possible by contacting the Sanitel-Med helpdesk. Data input can be automatic (through xml) or manual.
Sanitel-Med is linked to SANITEL, a database used for epidemiological surveillance and owned by the Belgian Federal Agency for the Safety of the Food Chain. From SANITEL, the farm capacity numbers of pigs and poultry are extracted to be used in the analysis of the AMU. For veal calves, monthly occupation numbers are obtained from SANITEL as well, by taking the average of the number of calves present at day 2, 11 and 21 of the month and day 2 of the consecutive month.
In pigs the usage of ZnO, authorised for prevention of diarrhoea in weaners, needs to be registered.
Analysis
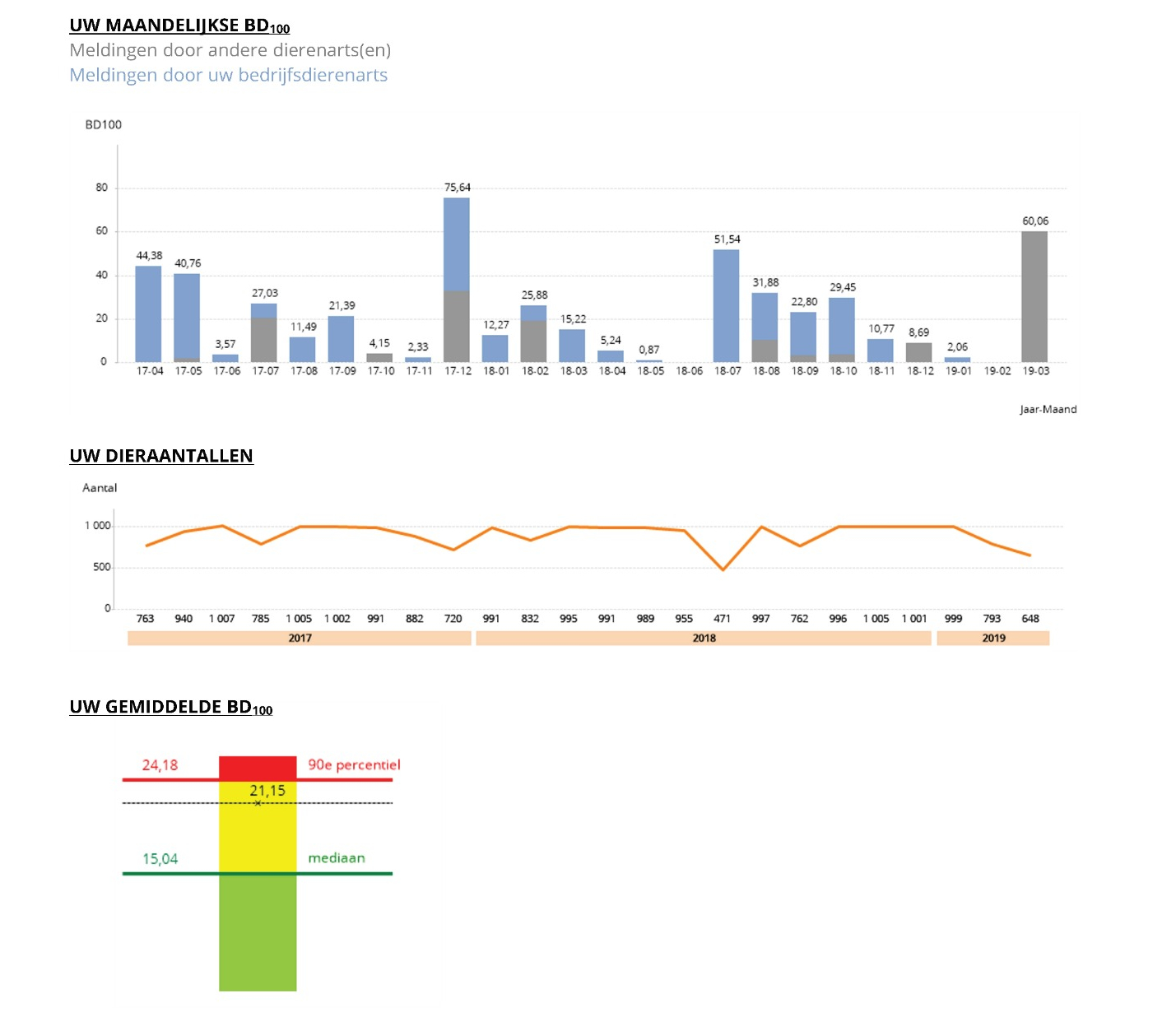
For benchmarking at farm-level, the dose-based unit of measurement DDDAbel (defined for Belgium at product-level) is used in the calculation of the indicator BD100 (treatment days per 100 days). To calculate the (kg) pigs and poultry at risk of treatment, standard weights proposed by EMA[1] are used (+ 2 kg for laying hens, agreed with the sector). Standard weights for veal are agreed with the sector. A BD100 per month is calculated, based upon which a yearly average BD100 is calculated per species and category (pigs and poultry). Farm-level results are also used for comparing sectors, assessing evolutions of AMU, and cross-check with national sales data. Therefore, the BD100-species is calculated, with the denominator corresponding to the average yearly number of pigs, broilers and veal calves per year and the numerator being the total number of DDDA-bel per species per species per year.
Benchmarking
Benchmarking (and reporting) at farm-level is done twice a year for pigs and poultry (per category) and veal calves, based on the average BD100. A ‘fixed benchmarking’ methodology is applied, with the results of each farm being compared with two threshold values (attention and action BD100) per weight category that are ‘fixed’ currently until 2023 as part of sector-specific reduction paths (https://amcra.be/nl/visie-2024/).
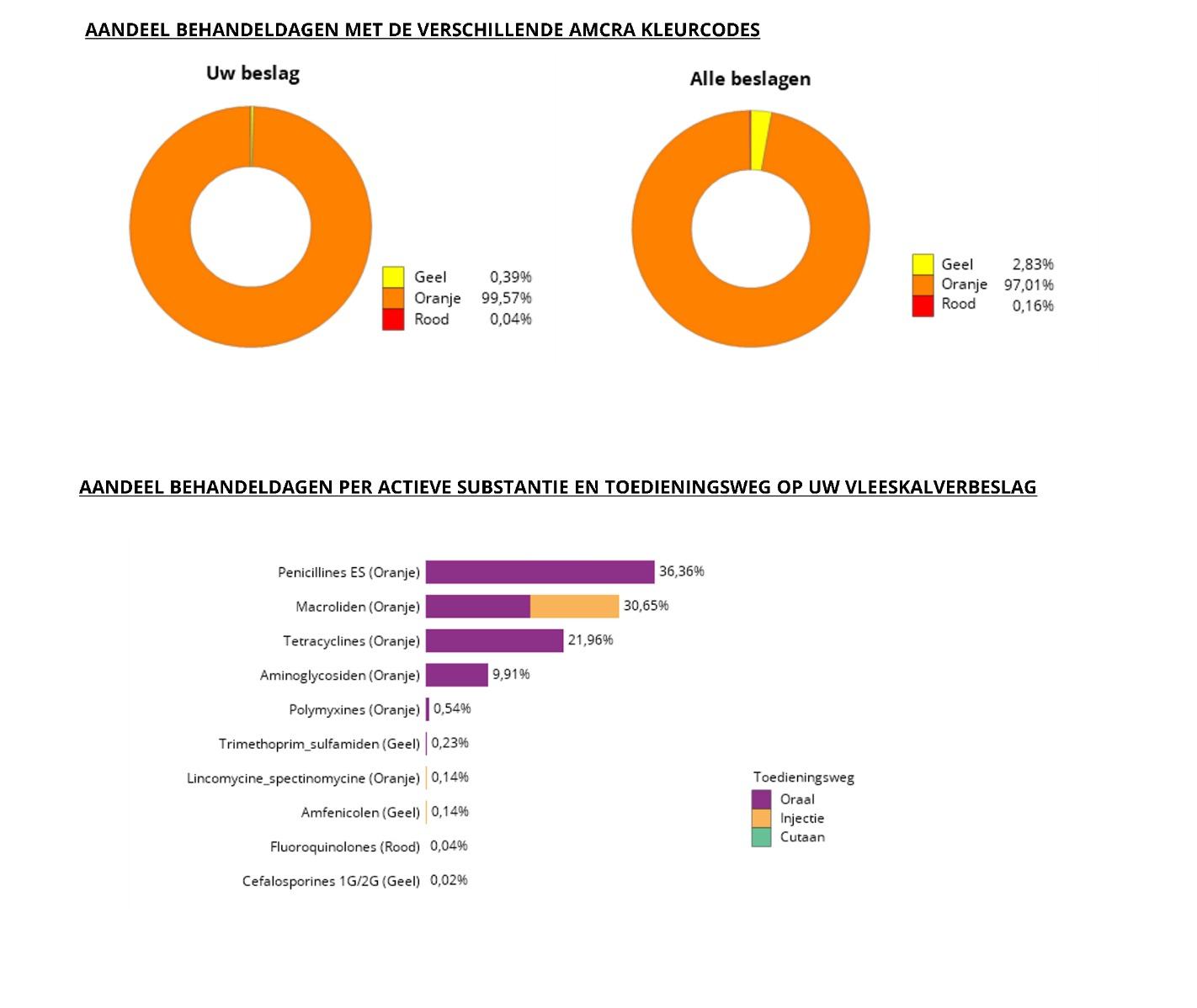
In addition, the type of antimicrobials used is benchmarked. Three colour codes of antimicrobials are distinguished: yellow, orange and red, the latter including the 3rd/4th gen. cephalosporins and the (fluoro)quinolones. The percentage of each antimicrobial class and each colour code in the total AMU in each weight category is compared to the mean percentages over all farms.
In pigs, use of premix is also benchmarked.
For benchmarking veterinarians, a contract score is calculated as a score out of 100 representing the ratio of animal-category-units, of farms where the vet is the responsible vet, that have green (low zone), yellow (medium = attention zone) or red (high = action zone) AMU. Based on the distribution of the scores of all veterinarian-animal species combinations, two threshold values are defined (the median and P90), dividing the vets in green, yellow and red vets.
Reporting
Veterinarians can access their benchmarking report 1x/year through the Sanitel-Med interface.
Below, examples are shown of the graphs included in a Sanitel-Med benchmarking report for veal calf farmers